Throughout the entire process of biopharmaceutical cell line development, "stability" has always been an unavoidable core issue—it is not an option, but a necessary prerequisite for determining whether a cell line can move from the laboratory to industrial production and realize commercial value. In many cases, high-expression clones selected in the laboratory, which seem to have high production potential, ultimately cannot be put into large-scale production due to insufficient stability, becoming "excellent products in the laboratory". Fundamentally, the ultimate goal of cell line development is to support industrial mass production, and stability is directly related to production reliability, product consistency, cost controllability, and regulatory passability. These four core needs together determine that stability is a mandatory question rather than a bonus question in cell line development, and the causal relationship between them runs through the entire process of biopharmaceutical industrialization.
Industrial production of biopharmaceuticals is essentially a long-term test of the cell line's ability to "continuously and stably produce qualified products". From seed bank construction, cell passage and expansion, to large-scale bioreactor culture and multi-batch continuous production, cells need to go through dozens or even hundreds of passages, and also bear multiple pressures such as fluctuations in culture environment, nutrient consumption, and accumulation of metabolic products. If the cell line lacks stability, any link that leads to loss of gene integration, silencing of target genes, decrease in expression level, or drift in product quality will result in loss of control of the production process, unqualified products, and even huge economic losses. Therefore, the reason why stability cannot be avoided in
cell line development is essentially determined by the "certainty demand" of industrial production—only a stable cell line can realize the repeatability and scalability of the production process, and convert the R&D achievements in the laboratory into large-scale commercial products.
Production Reliability: No Stability, No Possibility of Large-Scale Production

The core demand of industrial production is "stability, efficiency, and replicability", and the stability of the cell line is the primary prerequisite for achieving this demand, which is also one of the core reasons why cell line development must focus on stability. High-expression cell lines in the laboratory, if they can only maintain high output in short-term culture, but experience a sharp drop in expression level, decline in cell viability, or even abnormal proliferation and increased apoptosis rate during passage expansion or long-term culture, will directly lead to the failure of the production process to land.
In industrial production, cell lines need to go through multiple links such as seed expansion, working seed bank preparation, and batch culture in bioreactors, with the number of passages usually reaching dozens. If the cell line is genetically unstable, it may lead to the loss of target gene integration sites and chromosomal abnormalities, which in turn will cause significant fluctuations in the expression level of the target protein—for example, the original expression level of 1g/L drops to below 0.3g/L after more than a dozen passages, which will directly lead to the failure of the production batch to meet the output standard, and the production line cannot operate according to the standard process. More seriously, unstable cell lines may suddenly "stop working" during the culture process, leading to the scrapping of the entire culture batch, which not only wastes bioreactor operation time, culture medium and other raw materials, but also delays the production schedule and affects product supply.
It can be seen that stability is the "ballast stone" of industrial production. The reason why stability cannot be avoided in cell line development is precisely because only a stable cell line can ensure the continuity and repeatability of the production process, and turn large-scale production from "possible" to "realistic". If stability is ignored, even if high-expression clones are selected, they cannot support the long-term needs of industrial production and can only stay in the laboratory stage.
Product Consistency: No Stability, No Guarantee of Qualified Drugs
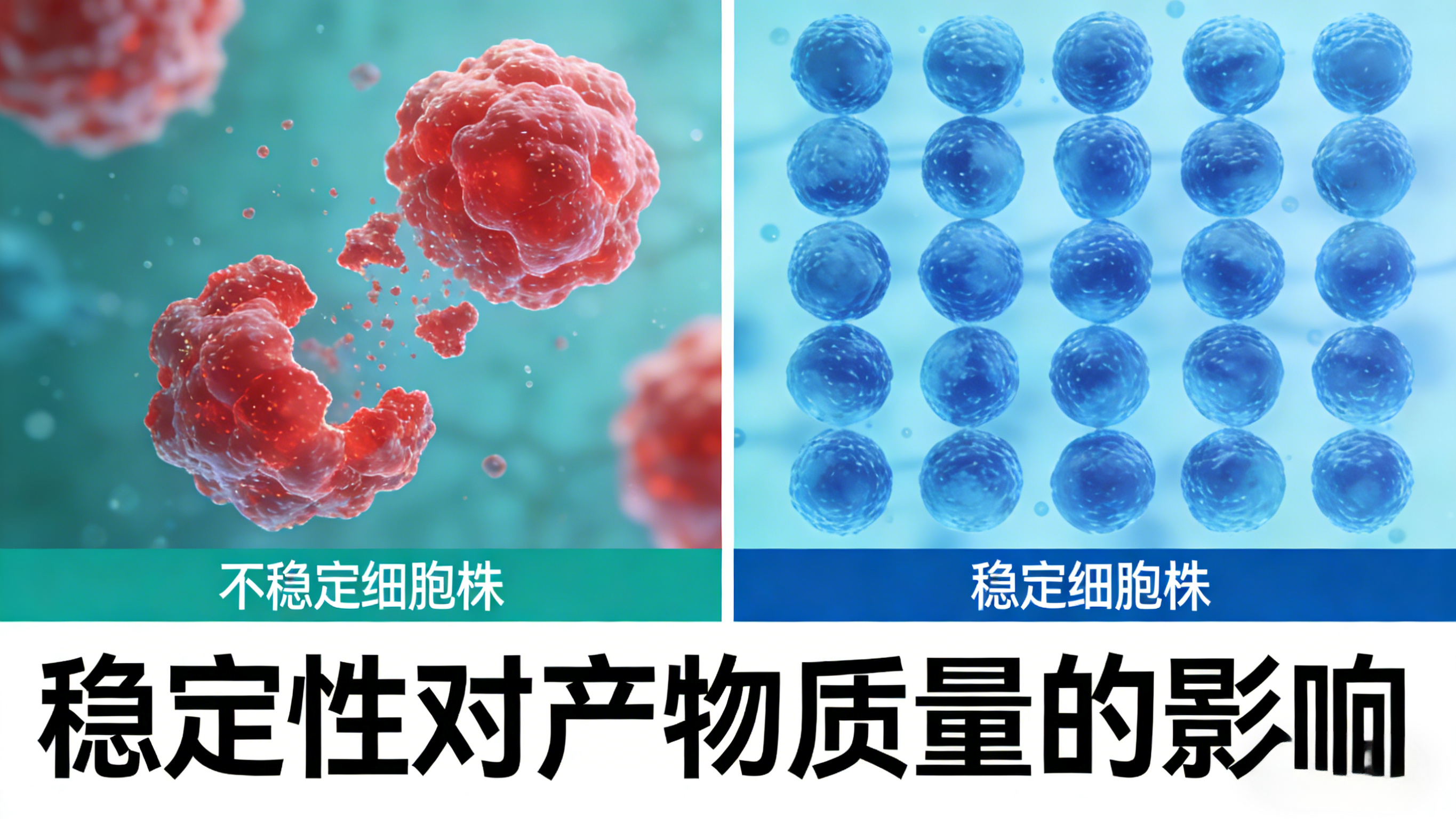
The quality of biopharmaceutical products (such as monoclonal antibodies, recombinant proteins, etc.) is directly related to clinical efficacy and drug safety, and the stability of the cell line is a key factor determining the consistency of product quality—which is also one of the core logics why stability cannot be avoided in cell line development. Unlike chemical pharmaceuticals, biopharmaceutical products are products of cell metabolism, and their quality attributes (such as glycosylation modification, aggregate content, charge heterogeneity, etc.) are highly dependent on the physiological state and expression environment of cells. The stability of the cell line directly determines whether cells can maintain a stable physiological state and expression characteristics during long-term production.
If the cell line is unstable, abnormal gene expression regulation during passage or culture may lead to changes in the structure of the target protein—for example, abnormal glycosylation modification may affect the half-life and immunogenicity of the protein, excessive aggregate content may cause adverse reactions, and excessive charge heterogeneity may lead to decreased efficacy. These quality abnormalities will not only result in unqualified products, but also bring potential safety risks to clinical medication. Industrial production requires that the quality of each batch of products remains highly consistent, and the realization of this consistency must be based on the stability of the cell line: only stable expression of the cell line can ensure that the quality attributes of each batch of products meet the standards, and ensure the clinical efficacy and safety of the drugs.
From the perspective of causal relationship, the stability of the cell line is the "cause", and the consistency of product quality is the "effect". The reason why stability cannot be avoided in cell line development is essentially to uphold the bottom line of drug quality and ensure that each batch of products can meet the standards for clinical use, which is also the core responsibility of the biopharmaceutical industry.
Cost Controllability: No Stability, No Commercially Feasible Value
The cost of industrial production of biopharmaceuticals is extremely high, involving multiple links such as cell line development, culture medium procurement, bioreactor operation, labor input, and quality testing. The instability of the cell line will directly lead to a significant increase in production costs, and even make the project lose commercial feasibility—which is also a practical consideration for cell line development to focus on stability.
On the one hand, the development cycle of
cell lines is long and the investment is large. The development of a commercial cell line often takes months or even years, involving a lot of clone screening and verification work. If the selected cell line has insufficient stability and cannot meet the needs of industrial production, it is necessary to re-screen clones and re-conduct process verification, which will not only extend the development cycle, but also waste all the previous investment in manpower, material resources and financial resources. On the other hand, in the process of industrial production, unstable cell lines will lead to an increase in the scrap rate of batches—each batch of bioreactor operation, culture medium consumption and labor input can be called "huge costs". Once the batch is scrapped, these investments cannot be recovered. In addition, unstable cell lines may require frequent adjustments of culture conditions and increased frequency of quality testing, which further increases the labor and material costs in the production process.
On the contrary, stable cell lines can effectively reduce production risks, reduce the scrap rate of batches, and ensure the continuity and efficiency of the production process. At the same time, stable cell lines do not require frequent adjustment of process parameters, which can simplify the production process and reduce quality testing costs, thereby realizing cost optimization of the entire industrial production process. Therefore, the reason why stability cannot be avoided in cell line development is an inevitable choice to achieve cost controllability and ensure the commercial feasibility of the project.
Regulatory Passability: No Stability, No Possibility of Product Launch

The launch of biopharmaceutical products must pass the strict approval of national drug regulatory authorities (such as China NMPA, US FDA, European EMA, etc.), and the stability study of cell lines is one of the core requirements for regulatory approval—which is also a rigid constraint that stability cannot be avoided in cell line development. Without a stable cell line, it is impossible to meet regulatory requirements. Even if the product quality meets the standards in the laboratory stage, it cannot be approved for launch and ultimately cannot be converted into commercial value.
National drug regulatory authorities clearly require that biopharmaceutical companies must provide sufficient cell line stability data when applying for new drugs, including passage stability, gene integration stability, target protein expression level stability, and product quality stability. For example, it is usually required that the cell line can maintain a stable gene integration state, expression level and product quality under the passage conditions simulating industrial production (such as passage ≥ 60 generations), to ensure that the quality of each batch of products in the production process is controllable. If qualified stability data cannot be provided, the drug regulatory authority will consider that the cell line cannot meet the requirements of industrial production and will not approve its launch.
This means that stability is the "passport" for cell lines to enter the market. The reason why stability cannot be avoided in cell line development is essentially determined by regulatory requirements, and it is a necessary condition for products to be successfully launched and realize commercial value. Ignoring stability research, even if high-expression clones are selected, they may fail to pass regulatory approval and end up in vain.
Conclusion: Stability Is the Core Bottom Line of Cell Line Development
In summary, the reason why stability cannot be avoided in cell line development is that there is a direct causal relationship between stability and the four core needs of industrial production (production reliability, product consistency, cost controllability, and regulatory passability)—stability is the cause, and the realization of the four needs is the effect. The ultimate goal of cell line development is never to simply pursue high expression, but to cultivate "stable and efficient" cell lines. Because only stable cell lines can support large-scale and standardized industrial production, ensure the stability and reliability of product quality, control production costs, meet regulatory requirements, and ultimately realize the commercial and clinical value of biopharmaceuticals.
In today's increasingly competitive biopharmaceutical industry, stability has become the core bottom line of cell line development. Ignoring stability is equivalent to giving up the feasibility of industrial production and the opportunity of product launch. Therefore, in every link of cell line development—from clone screening and gene integration to passage verification and process optimization—stability should be taken as the core indicator, continuously concerned and strictly controlled. Only in this way can we cultivate cell lines that meet the needs of industrial production and promote the high-quality development of the biopharmaceutical industry.



