Cell line construction is a core underlying technology in fields such as biopharmaceuticals, recombinant protein production, and gene function research. Especially in the industrial development of therapeutic monoclonal antibodies, bispecific antibodies, fusion proteins, and recombinant vaccines, it directly determines the production capacity, quality, and production cost of products. In recent years, with the continuous expansion of the biopharmaceutical market demand, the industry's core demands for cell lines have increasingly focused on two key indicators: first, high expression level, i.e., the output efficiency of target proteins per cell and per unit volume, which is directly related to industrial production capacity and cost control; second, long-term stability, i.e., the ability of the cell line to maintain gene integrity, expression level consistency, and protein quality homogeneity during continuous passage and large-scale culture, which is a core prerequisite for drug approval and large-scale production. Currently, the Chinese Hamster Ovary (CHO) cell-based mammalian cell expression system dominates the commercial market, while cell lines such as HEK293 are mostly used for rapid expression in the R&D stage. The industry's technological iteration has always focused on solving the industry pain point of "difficulty in balancing high expression and stability". This article focuses on these two core factors to sort out the current development status, key technologies, and existing challenges of cell line construction.

I. Core Technologies and Current Development Status of High-Expression Cell Line Construction
High expression level is the core competitiveness of industrial application of cell lines. Early traditional cell line construction technologies mostly relied on random integration and extensive screening, with expression levels only reaching the milligram level, which was difficult to meet the needs of industrial production. After decades of technological iteration, the industry has now formed a full-chain optimization system from vector design, gene integration, host modification to high-throughput screening. The expression level of recombinant proteins has increased from less than 1g/L in the early stage to 10-15g/L currently, and some optimized cell lines even exceed 20g/L. The core technological breakthroughs are concentrated in the following dimensions:
1. Rational Optimization of Expression Vector Elements to Improve Transcription and Translation Efficiency from the Source
The
expression vector is the core carrier for regulating the expression of exogenous genes, and its element combination directly determines the expression intensity. Currently, the industry has shifted from blind splicing to rational design. Screening and modification of strong promoters are the core links. Although the traditional CMV promoter has high expression intensity, it is prone to epigenetic silencing. At present, the industry mostly adopts modified CMV, EF1α, SV40 and other composite strong promoters, or endogenous promoters adapted to host cells, to balance transcriptional activity and sustainability; at the same time, regulatory elements such as enhancers, insulators, and matrix attachment regions (MAR) are added to break the inhibition of chromatin structure and improve transcription efficiency.
Secondly, codon optimization and translation element optimization are crucial. According to the codon preference of host cells such as CHO, synonymous mutations are performed on the target gene sequence to eliminate rare codons and secondary structures, thereby greatly improving mRNA translation efficiency; at the same time, optimizing the Kozak sequence and signal peptide sequence to enhance protein secretion efficiency, reduce intracellular protein accumulation-induced cytotoxicity, and further improve the effective expression level. In addition, the application of bicistronic expression elements (such as IRES and 2A peptides) realizes the co-expression of target genes and screening markers, improving the enrichment efficiency of positive clones.
2. Upgrade of Gene Integration Strategies to Achieve Efficient Site-Directed Integration and Precise Copy Number Regulation
Traditional random integration technology is a core bottleneck restricting the expression level. Exogenous genes inserted randomly into the silent regions of the host genome not only have extremely low expression levels but also have strong clonal heterogeneity. Currently, the industry has gradually eliminated extensive random integration and turned to semi-targeted integration (STI) and site-specific integration (SSI) technologies. Among them, transposon-mediated semi-targeted integration (such as PiggyBac system) can directionally insert exogenous genes into transcriptionally active regions, increasing integration efficiency several times, greatly improving cell pool homogeneity, and significantly shortening the screening cycle; the CRISPR/Cas9-based homology-directed repair technology realizes precise single-copy integration of exogenous genes at the "safe harbor" sites of the genome, completely avoiding expression differences caused by position effects, and the single-copy expression efficiency is 3-5 times higher than that of random integration.
At the same time, the optimization of the screening marker system helps precise regulation of copy number. The DHFR/MTX and GS/MSX screening systems achieve controllable amplification of exogenous gene copy number by gradually increasing the screening pressure, which not only ensures high expression level but also avoids excessive metabolic burden caused by over-amplification, balancing the relationship between cell growth and protein expression.
3.
Host Cell Engineering Modification to Optimize Cell Growth and Metabolic Characteristics
The physiological state of host cells directly affects the expression efficiency. Currently, the industry uses gene editing technology to directionally modify host cells such as CHO to improve cell viability, anti-apoptotic ability, and metabolic efficiency. For example, knocking out pro-apoptotic genes such as BAX/BAK to extend the cell culture cycle; overexpressing chaperone proteins to alleviate endoplasmic reticulum stress and improve the efficiency of correct protein folding; optimizing glycosylation-related genes in cells to balance high expression and protein post-translational modification quality. At the same time, the popularization of serum-free suspension-adapted host cells is compatible with large-scale bioreactor culture, further releasing the high expression potential.
4. High-Throughput Intelligent Screening to Rapidly Enrich High-Producing Clones
The traditional limiting dilution method has a long screening cycle and low efficiency, making it difficult to quickly screen high-producing clones. Currently, the industry generally adopts flow cytometry sorting (FACS), single-cell printing, high-throughput well plate culture combined with fluorescent reporter gene technology to achieve rapid enrichment of high-expression cells, shortening the screening cycle from 8-12 weeks to 3-4 weeks; combining artificial intelligence and metabolomics analysis to predict the expression level through cell morphology and metabolic parameters, further improving the accuracy of high-producing clone screening and avoiding missing high-quality cell lines.
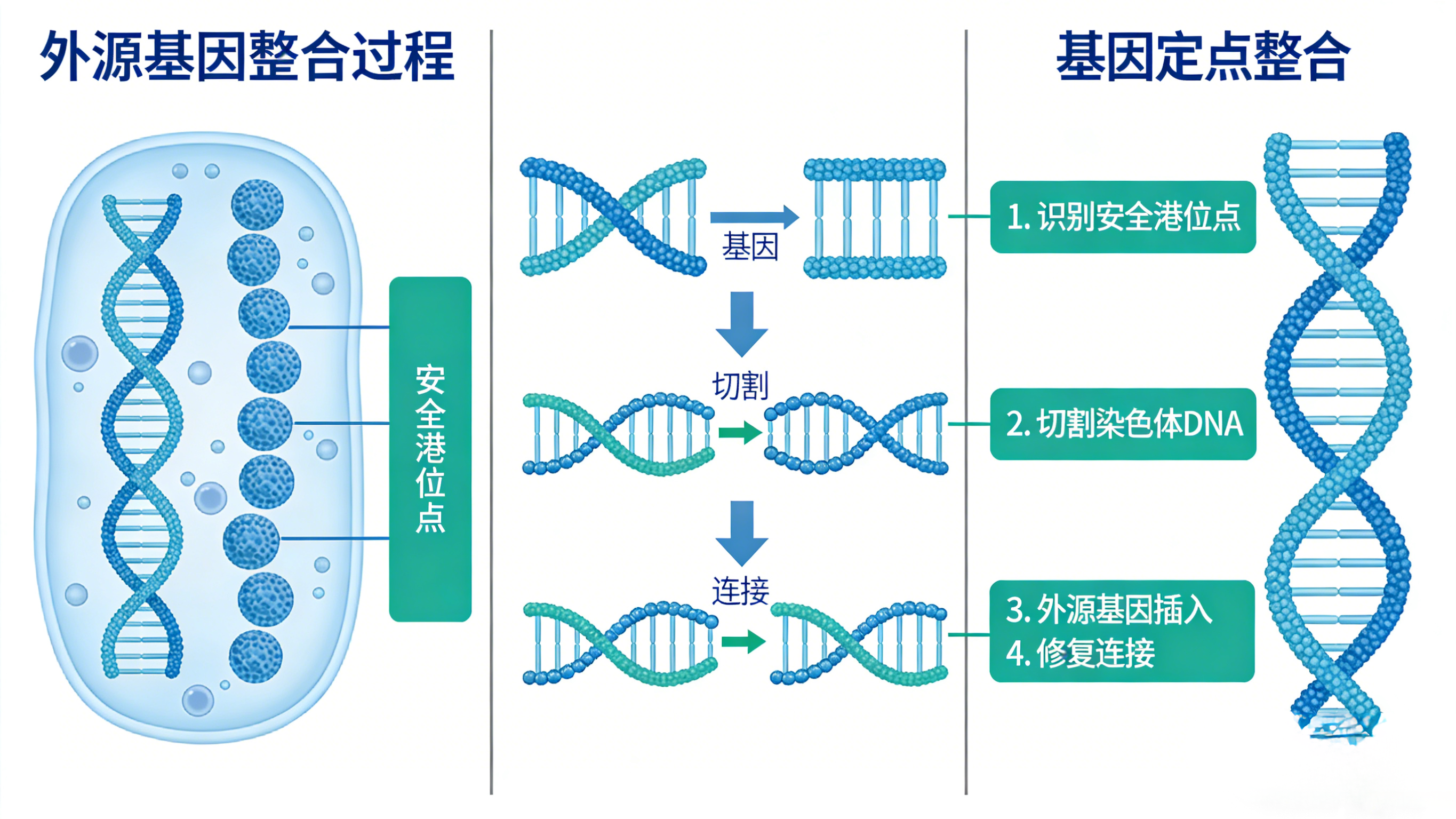
II. Core Influencing Factors and Technical Optimization Progress of Cell Line Stability
Stability is a core prerequisite for cell lines to have industrial value, which is divided into genetic stability and expression stability. The former refers to the stability of exogenous gene integration sites and copy numbers during continuous passage, while the latter refers to the fluctuation of expression level within ±15%-20% after long-term passage of the cell line (usually requiring 60-90 passages), and no obvious drift in protein quality. Early high-producing cell lines often had problems such as "sharp drop in yield after passage", "gene silencing", and "chromosomal variation", which seriously restricted industrial application. Currently, the industry has made breakthrough progress in technical optimization for stability.
1. Core Influencing Factors: Key Pain Points Restricting Stability
First, position effect and epigenetic silencing. Exogenously integrated genes randomly inserted into heterochromatin regions are prone to DNA methylation and histone modification during long-term culture, leading to transcriptional silencing and gradual attenuation of expression level; second, loss of gene copy number. Over-amplified exogenous genes are prone to fragment loss during cell division, especially the copy number decreases significantly after continuous passage, directly leading to reduced yield; third, host cell chromosomal instability. Engineering cells such as CHO themselves have problems such as chromosomal number variation and translocation, and karyotype instability occurs during long-term suspension culture, leading to cell population differentiation and a decrease in the proportion of high-producing clones; fourth, imbalanced cell metabolic burden. High expression leads to excessive cell metabolic pressure, decreased growth vitality, and indirectly affects expression stability.
2. Core Technical Strategies to Improve Stability
(1) Site-Directed Integration Technology to Avoid Position Effect from the Root
Targeted integration into genomic safe harbor sites is the core method to improve stability. Such sites do not interfere with the expression of essential genes of host cells, have an open chromatin structure, can continuously support exogenous gene transcription, and completely avoid epigenetic silencing caused by random integration. Tests have shown that after continuous passage of site-directed integrated cell lines for more than 90 days (26 passages), the expression level fluctuation is controlled within 20%, and the gene copy number has no obvious change, which is far superior to randomly integrated cell lines and has become the mainstream construction strategy for commercially produced cell lines.
(2) Optimization of Vector Elements to Resist Epigenetic Silencing
Adding MAR elements and insulator sequences to the expression vector to block the inhibitory signals of surrounding chromatin and maintain gene transcriptional activity; modifying the promoter sequence to reduce the probability of methylation sites, and selecting endogenous promoters of host cells to reduce the silencing risk of exogenous promoters. At the same time, low-copy precise integration is used instead of high-copy random amplification to avoid gene fragment loss, balance expression level and genetic stability, and solve the industry problem of "high yield but unstable".
(3) Host Cell Stability Modification to Enhance Genetic Robustness
Using gene editing technology to repair host cell chromosomal defects, knock out genes related to abnormal recombination, and improve chromosomal division stability; screening host cell subclones with stable karyotype as the chassis cells for cell line construction, reducing the risk of population differentiation from the source. At the same time, optimizing the cell culture process, using serum-free chemically defined medium, combined with dynamic feeding strategies, to reduce the cell metabolic burden, maintain the stable physiological state of cells, and indirectly ensure the continuity of expression.
(4) Strict Stability Verification System to Adapt to Industrial Standards
Currently, the industry has established a standardized stability verification process, requiring continuous passage of cell lines for more than 60 generations, regular detection of gene copy number, mRNA expression level, protein yield and quality indicators, and verification of genetic integrity through karyotype analysis, Southern blot and other technologies. Regulatory authorities have increasingly strict requirements on the stability of production cell lines. Cell lines with insufficient stability period and excessive expression fluctuation cannot be approved for industrial production, forcing technological iteration towards the direction of "high yield and long-term stability".

III. Current Technical Bottlenecks and Cutting-Edge Development Trends
1. Existing Core Bottlenecks
Although significant breakthroughs have been made in high expression level and stability technologies, there are still some industry pain points: first, although site-directed integration technology has excellent stability, the expression intensity of some sites still has room for improvement, and the construction cost is high and the process complexity is increased; second, for the construction of cell lines for special proteins (such as complex glycosylated proteins and toxic proteins), it is difficult to balance high expression, stability and protein activity at the same time; third, the adaptability difference between small-scale screening and large-scale bioreactor culture, some laboratory high-producing cell lines have decreased stability and reduced expression level in large-scale production; fourth, the problem of clonal heterogeneity has not been completely eliminated, even with site-directed integration, there are still a small number of cell population expression differences.
2. Cutting-Edge Development Trends
In the future, cell line construction technology will develop towards precision, intelligence, and standardization, and the in-depth integration of synthetic biology and artificial intelligence will become the core driving force. On the one hand, through modular vector design and multi-gene co-regulation, cell lines with adaptive metabolic balance will be constructed to achieve perfect compatibility between high expression and stability; on the other hand, AI-assisted rational design of promoters and signal peptides will greatly improve the efficiency and success rate of element optimization; at the same time, one-time integration and screening marker-free cell line construction technologies will simplify the process and reduce regulatory risks; in addition, the development of universal chassis cell lines will realize rapid adaptation of different target proteins, shorten the cell line development cycle, and adapt to the market demand for rapid iteration of biopharmaceuticals.
IV. Conclusion
Cell line construction technology has evolved from early extensive random construction to a new stage of precision and rational design. High expression level and stability are no longer conflicting indicators, but are synergistically improved through technologies such as vector optimization, site-directed integration, host modification, and intelligent screening. Currently, CHO cells are still the core host for commercial production, site-directed integration and gene editing technologies have become the industry mainstream, and the expression level and stability indicators of cell lines have continued to break through, greatly reducing the production cost of biopharmaceuticals and improving the homogeneity of product quality.
In the future, with the continuous iteration of technology, cell line construction will further break through the existing bottlenecks, realizing the upgrade from "high yield" to "high and stable yield", and from "laboratory adaptation" to "full-scale production adaptation", providing a more solid underlying support for the R&D and industrialization of innovative biopharmaceuticals, and at the same time promoting the biopharmaceutical industry to develop in the direction of high efficiency, low cost, and standardization.


